The molecular formula for water is H2O. One molecule of water consists of one oxygen atom covalently bonded to two hydrogen atoms.
There are three isotopes of hydrogen. The usual chemical formula for water assumes the hydrogen atoms consist of the isotope protium (one proton, no neutrons). Heavy water is also possible, in which one or more of the atoms of hydrogen consist of deuterium (symbol D) or tritium (symbol T). Other forms of the chemical formula for water include D2O, DHO, T2O, and THO. It's theoretically possible to form TDO, although such a molecule would be extremely rare.
Although most people assume water is H2O, only completely pure water lacks other elements and ions. Drinking water usually contains chlorine, silicates, magnesium, calcium, aluminum, sodium, and trace amounts of other ions and molecules.
Also, water dissolves itself, forming its ions, H+ and OH-. A sample of water contains the intact water molecule along with hydrogen cations and hydroxide anions.
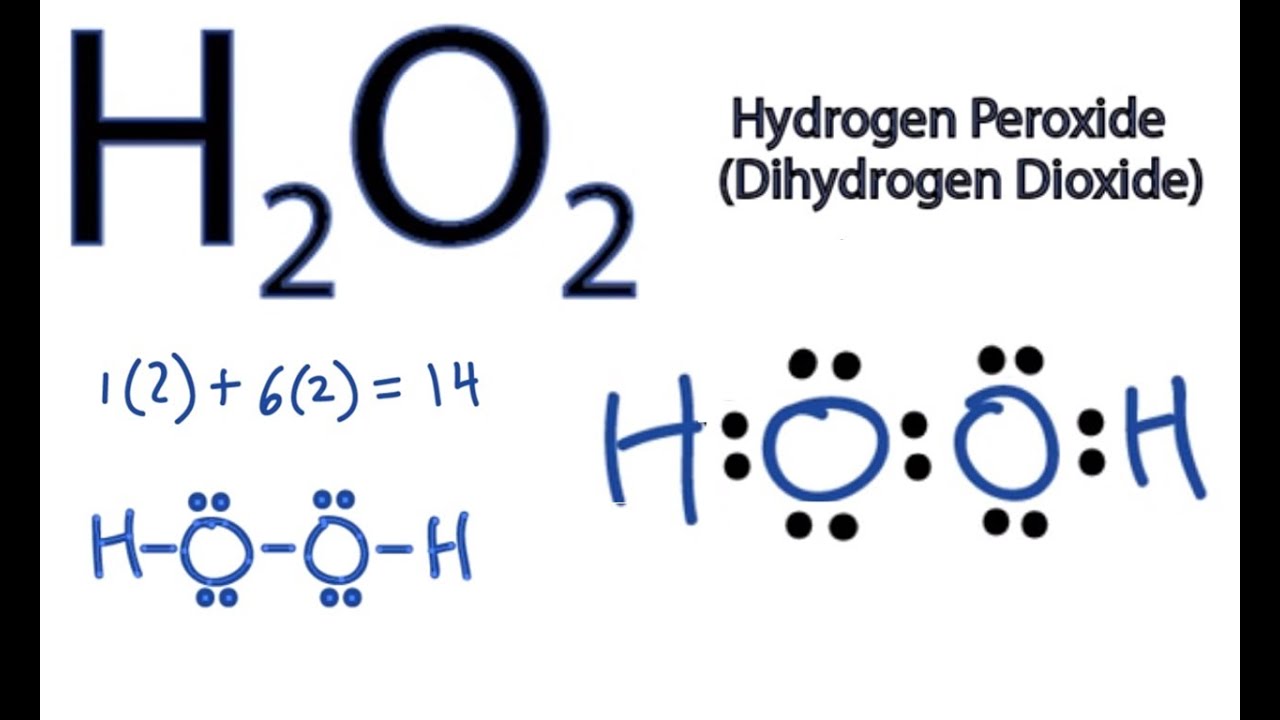
Explore this ArticleWriting Chemical Formulas of Covalent CompoundsWriting Chemical Formulas of Ionic CompoundsDetermining the Products Given ReactantsArticle SummaryQuestions & AnswersRelated ArticlesReferences
The molecular formula is just each element of the molecule and the number of each of these elements, while the structural formula is when the structure is drawn out individually For example glucose molecular formula is C6H12O6, but its structural formula is as follows As you can see the structural formula shows the bonding and structure of each molecule and each individual element. Clicking on a first atom and on a second atom. You may also paste a molfile or a SMILES by moving your pointer over the structure editor and press 'CTRL + V'.
This article was co-authored by Meredith Juncker. Meredith Juncker is a PhD candidate in Biochemistry and Molecular Biology at Louisiana State University Health Sciences Center. Her studies are focused on proteins and neurodegenerative diseases.
There are 15 references cited in this article, which can be found at the bottom of the page.
There are 15 references cited in this article, which can be found at the bottom of the page.
A good way to think about a chemical reaction is the process of baking cookies. You mix the ingredients together (flour, butter, salt, sugar, and eggs), bake it, and see that it changes into something new: cookies! In chemistry terms the equation is the recipe, the ingredients are 'reactants,' and the cookies are 'products.' All chemical equations look something like 'A + B →C (+ D...),' in which each letter variable is an element or a molecule (a collection of atoms held together by chemical bonds). The arrow represents the reaction or change taking place. Some equations may have a double-headed arrow (↔), which indicates that the reaction can proceed either forward or backward. To write the equations there are a number of important naming rules that you need to know.
